Company updates and highlights
Stay informed about FOMAT initiatives, new capabilities, awards and other key news.
FOMAT Acquires Topography Health: A Conversation With CEO Nick Focil
FOMAT acquires Topography Health — and the story behind the deal started with a dinner hint at the MAGI Clinical Research Conference. At the recent MAGI Clinical Research Conference, Clinical Leader editor Dan Schell crossed paths with Nick Focil, CEO of...
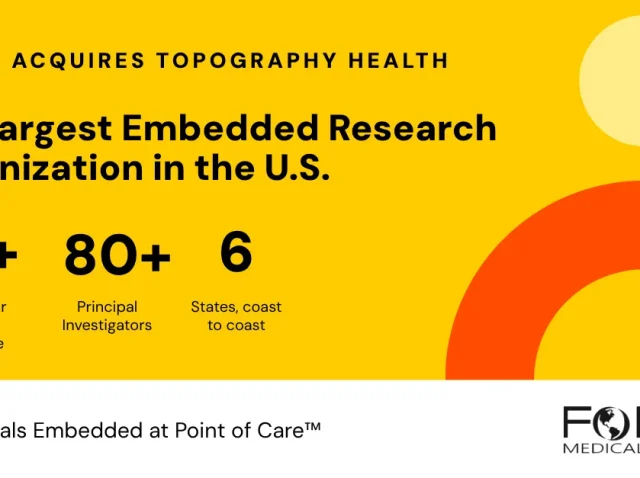
FOMAT Acquires Topography Health and Becomes the Largest Embedded Research Organization in the U.S.
Clinical research has long been concentrated in large academic medical centers and specialized facilities — places that most patients never visit. FOMAT Medical Research was built on the belief that this needed to change. On May 1, 2026, that mission reached...
Hispanic Community Perspectives on Clinical Trial Participation
Hispanic clinical trial participation remains critically low despite being one of the fastest-growing demographic groups in the U.S. This gap not only limits the generalizability of research findings, but also deepens existing healthcare disparities. Hispanic individuals make up nearly 19% of...
Expanding Ophthalmology Clinical Trials in Central California
FOMAT is expanding its ophthalmology clinical trials network in Central California through a new partnership with Dr. Daniel R. Agarwal, an ophthalmologist specializing in retina and vitreous care at San Luis Obispo Eye Associates. This collaboration reflects FOMAT’s commitment to bringing...
FOMAT Medical Research Expands to Santa Maria, California, Strengthening Regional Access to Clinical Research
FOMAT has opened a new hub, making clinical research in Santa Maria, California more accessible than ever for underserved communities in the region. Building on years of active research presence in the area, this expansion strengthens the organization’s capacity to connect...
New Partnership Between FOMAT Medical Research and Golden Valley Health Centers Brings Clinical Trials to North-Central California’s Diverse Populations
Clinical Trials Diverse Communities: FOMAT and Golden Valley Health Centers Partner Clinical trials diverse communities across North Central California will now have access to through a new partnership between FOMAT and Golden Valley Health Centers (GVHC). Announced on December 17, 2024,...
FOMAT Medical Research Wins 2024 Excellence in Patient Centricity Award
Patient Centricity Award 2024: FOMAT Wins SCRS Recognition The patient centricity award for 2024 has been earned by FOMAT, receiving the prestigious Excellence in Patient Centricity Award from the Society for Clinical Research Sites (SCRS). Sponsored by Sanofi, this award recognizes...
Ventura County Biotech Trade Mission to India: Fostering Global Growth and Innovation
The biotech trade mission to India led by Ventura County marks a significant step in connecting US based life science organizations with one of the world’s fastest growing biotechnology markets. Led by Ventura County Supervisor Jeff Gorell and organized by Brent...
FOMAT Medical Research Launches Community-Based Oncology Trials in Ventura and Surrounding Counties
FOMAT is a leading oncology clinical trial site California, now expanding its research portfolio with a dedicated oncology unit at its headquarters in Oxnard, Ventura County. This initiative brings community based cancer trials directly to local patients, offering access to innovative...