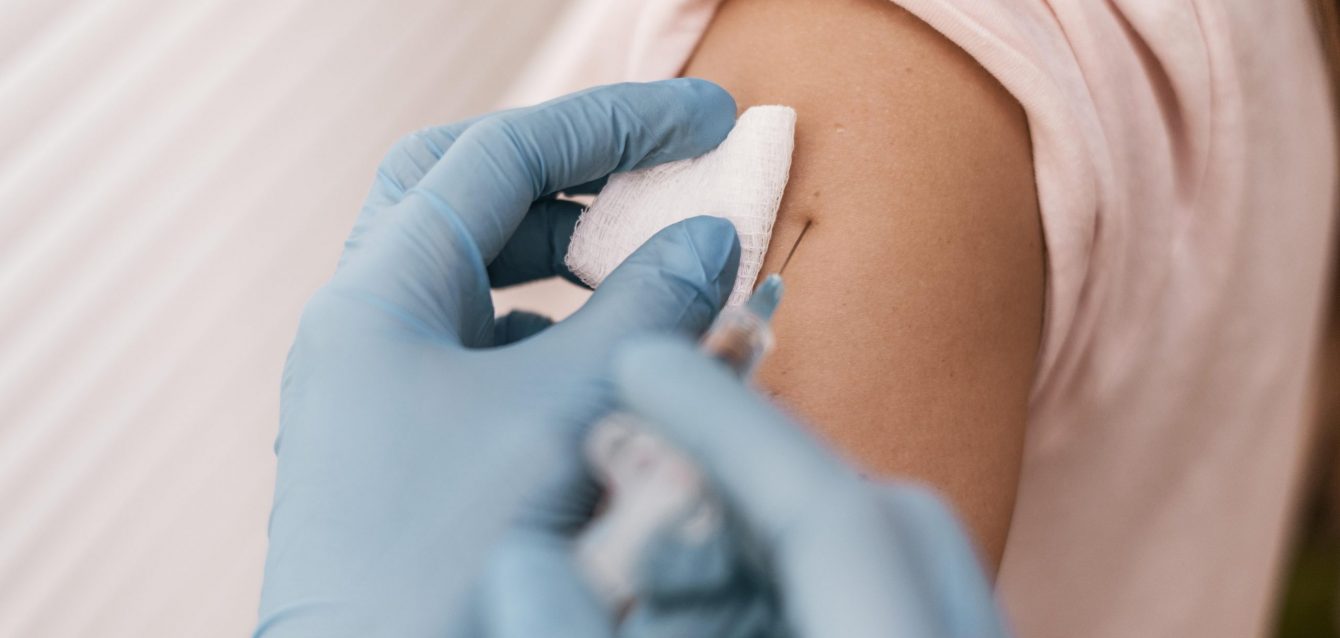
One of the most rapidly fast-tracked vaccines in history—an anti-Ebola “ChAd3” vaccine—just entered clinical trial in humans. It may wrap in November. It protected macaques for five weeks, leaping to 10 months when followed by a booster two months later. It…
FDA: Little Evidence to Support Testosterone Drugs
The Food and Drug Administration says there is little evidence that testosterone-boosting drugs taken by millions of American men are beneficial, though the agency is also unconvinced by studies suggesting the hormone carries serious testosterone therapy risks. The agency posted its…
Genetics to Test Ebola Vaccine
An Iowa drug developer is preparing to test a possible Ebola vaccine in humans, as scientists race to develop ways to prevent or fight a virus that has killed more than 1,000 people in a West African outbreak. NewLink Genetics is…
Debate Continues Over Experimental Ebola Drugs
In this photo taken on Tuesday, Aug. 12, 2014, Guinea soldiers stand around a rope across the road that separates Guinea and Sierra Leone, and works as a makeshift border control checkpoint at Gbalamuya-Pamelap, Guinea. As Guinea closed its border with…
Answers about an Unproven Drug for Ebola
Marie-Paule Kieny, Assistant Director General of the World Health Organization,WHO, informs the media following a panel of medical analysts to explore the ethics of experimental treatment in the Ebola outbreak, at the headquarters of the WHO in Geneva, Switzerland, Tuesday, Aug….